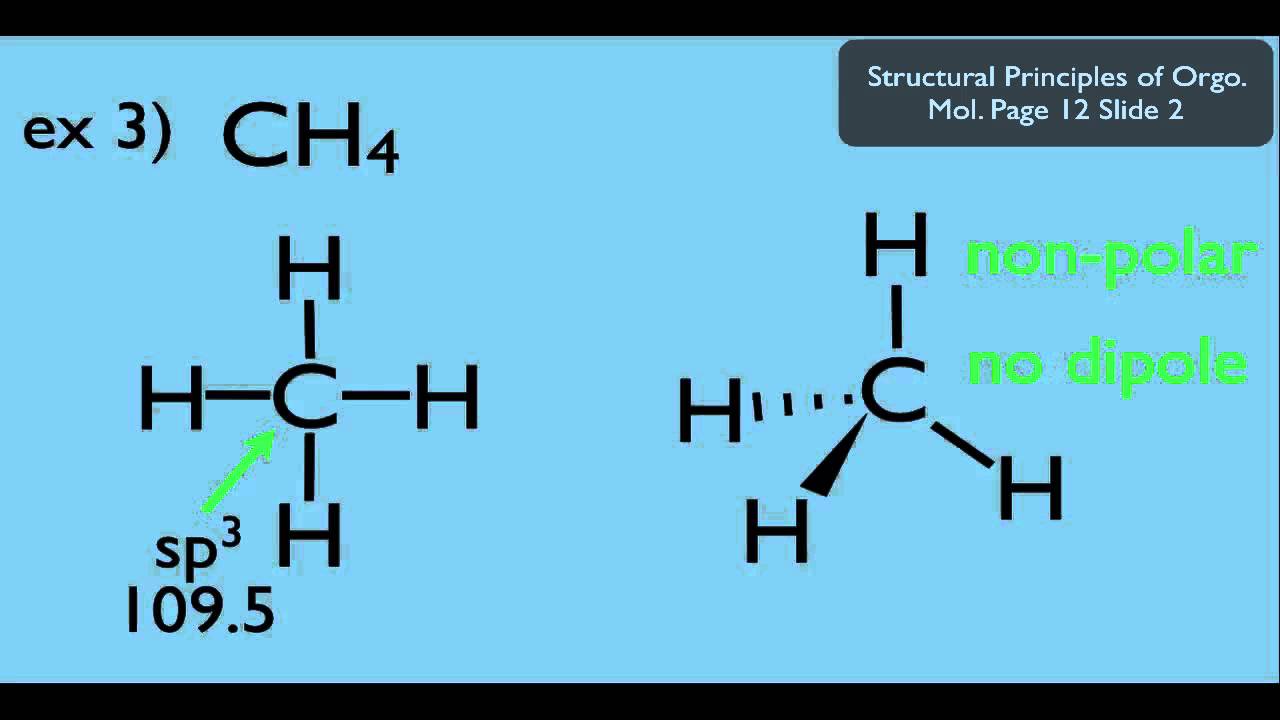
The difference between the electronegativity values of B and F is equal to 1.94 (3.98 – 2.04 = 1.94) which is greater than 0.5. To determine if a bond is polar or not we must find out the difference between the electronegativity values of the two atoms sharing a bond. The electronegativity of B (Boron) is 2.04 and of F (Fluorine) is 3.98 (maximum) according to the Pauling scale which means that F (Fluorine) will pull the shared electrons towards itself and will thus acquire a partial negative charge (δ-) and B (Boron) will have a partial positive charge (δ+). In BF3 (Boron Trifluoride) molecule there are three bonds of B-F, as shown clearly in Lewis diagram above. We can see that each F (Fluorine) atom has 3 lone pairs of electrons thus the molecular structure is balanced and symmetric.Īccording to the VSEPR (Valence Shell Electron Pair Repulsion) Theory, the BF3 (Boron Trifluoride) molecule has a Trigonal planar geometry with each F-B-F bond angle equal to 120 degrees which again adds to the balance of the molecule making it highly symmetric, as shown:Įlectronegativity & Bond Polarity of BF3 (Boron Trifluoride) The valency of B (Boron) is 3 and of F (Fluorine) is 7, thus the Lewis structure of BF3 can be drawn as shown in the figure: The BF3 (Boron Trifluoride) molecule has 1 atom of B (Boron, atomic no: 5) and 3 atoms of F (Fluorine, atomic no: 9). 
Uses of BF3 (Boron Trifluoride) Molecular Structure of BF3 (Boron Trifluoride)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
